In vitro characterization and cellular uptake profiles of TAMs-targeted lipid calcium carbonate nanoparticles for cancer immunotherapy
Abstract
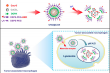
Tumor-associated macrophages (TAMs) are one of the key contributors to tumor development, accelerated tumor invasion and metastasis, and induction of immunosuppression. Targeted delivery of immunomodulatory agents to promote polarization of TAMs may alleviate the immunosuppressive tumor microenvironment. Calcium carbonate nanoparticles (CCN), which exhibit excellent biocompatibility, pH sensitivity, and easy surface modification, have attracted much attention in targeted nano delivery. In this study, CCN was used as the matrix material to develop a UNO peptide-modified lipid CCN for targeted immunomodulation of TAMs by using the mannose receptor overexpressed on the surface of TAMs as targets. The preparation of CCN was optimized by the single-factor test using the gas diffusion method with the particle size as the index. The surface modification of CCN with UNO peptide-modified phospholipids was performed and its targeting effect on TAMs was investigated. The average particle size of the CCN and UNO peptide-modified CCN was 144.5 ± 3.8 nm and 167.0 ± 1.3 nm, respectively. UNO peptide-modified CCN could enter TAMs via actively targeted uptake mediated by mannose receptors. These results demonstrated that the developed UNO peptide-modified CCN with controlled nano-size and excellent TAMs targeting properties is a highly promising nanocarrier for targeted delivery of TAMs immunomodulatory agents.